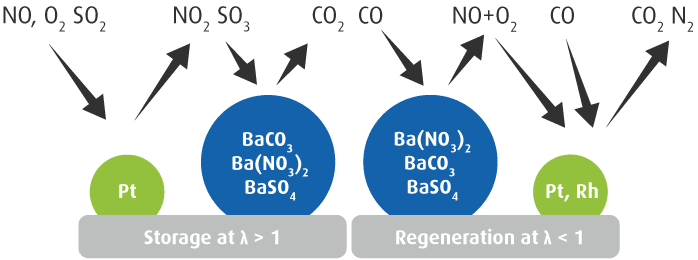
NOₓ Storage Catalyst (NSC)
The first step of the storage mechanism is the oxidation of NO to NO2 on the precious metals of the catalyst, mainly Platinum (Pt). This NO2 is subsequently stored on the corresponding storage materials (e.g., Barium Carbonate BaCO3) incorporated in the catalyst formulation until the desired storage capacity is reached.
The NSC is operated non-continuously, i.e., it needs to be regenerated periodically by means of rich exhaust gas (λ<1) in order to decompose the nitrates and reduce them.